Trillions of different microorganisms reside in our gut, known as the gut microbiota. The microbiota has emerged as a very important environmental factor regulating a wide range of host physiological processes, including metabolism, immune functions and circadian rhythms. The goal of our research is to identify the molecular basis of interactions between the microbiota and the circadian rhythms in mammalian metabolism and immunity. We use various techniques including genomics, gnotobiotics, laser capture microscopy to delve deeper into the transcriptional and epigenetic mechanisms that regulate host-microbial interactions. We anticipate that these efforts will lead to important new insight into how intestinal microbes regulate host physiological functions, and identify new avenues for developing therapeutics against metabolic and immunological diseases of the intestine.
Microbial regulation of circadian rhythms in the intestine
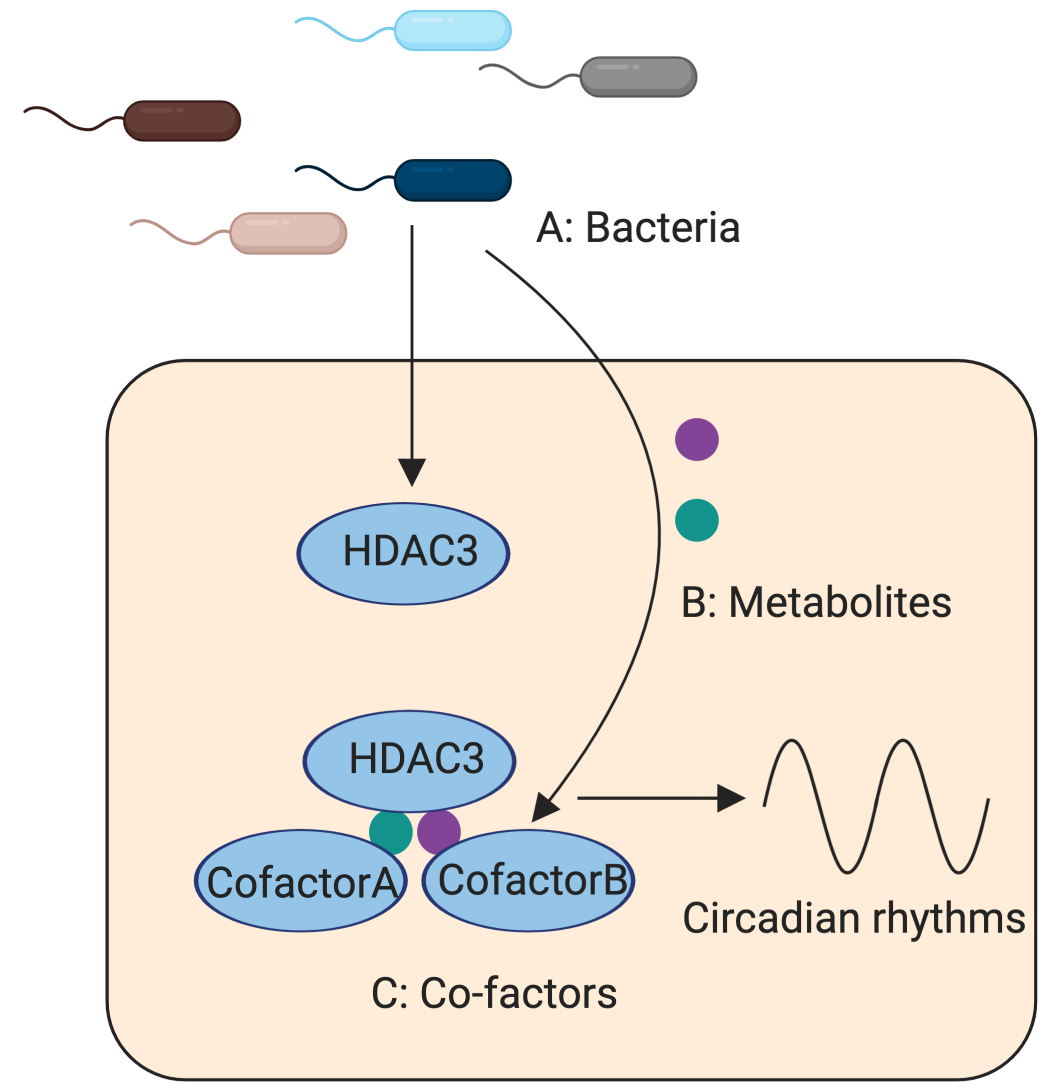
1. Identify regulatory factors that interact with intestinal HDAC3.
HDAC3 is a histone deacetylase that removes acetylation on histones and represses transcription. It also deacetylates non-histone proteins and regulates their functions. We have identified HDAC3 as a key molecule integrating microbial and circadian cues in the gut. In ongoing work, we are investigating how gut bacteria regulate HDAC3 and how HDAC3 controls various circadian programs to impact metabolic health and immune responses.
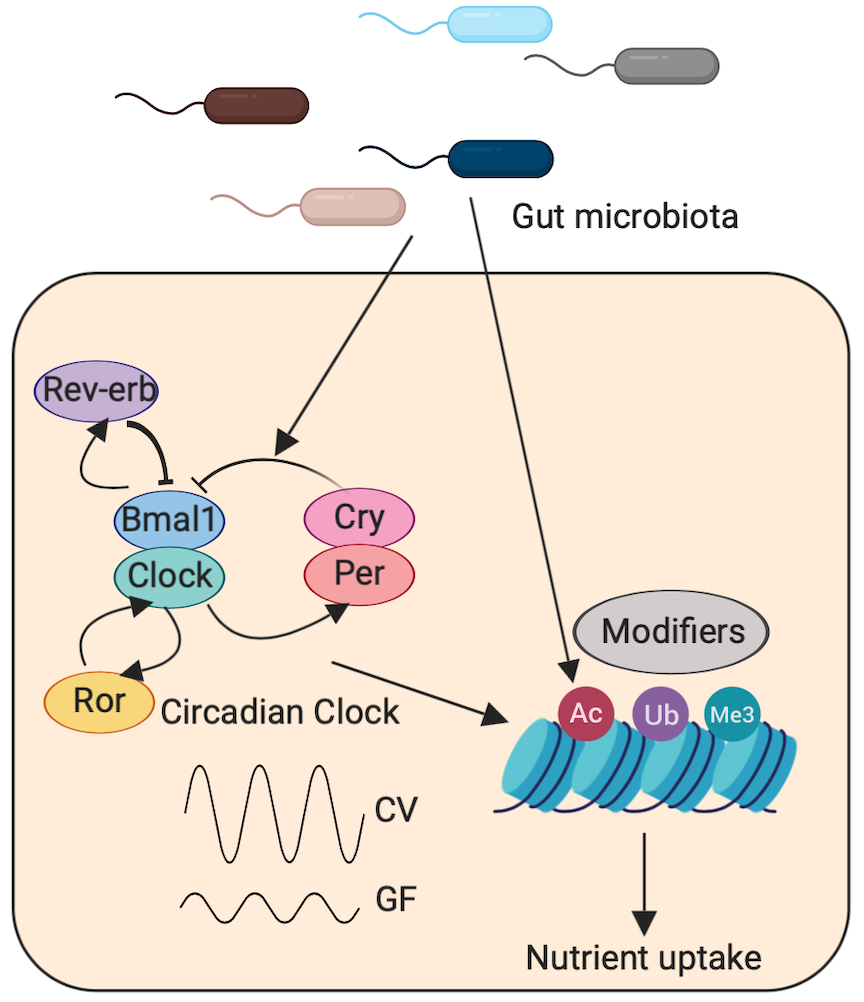
2. Determine epigenetic mechanisms underlying the cross-talk between the microbiota and circadian pathways.
Mammalian metabolic immune activities and immune responses are temporal orchestrated in accordance with diurnal sleep-wake and feeding-fasting cycles. It is becoming clear that the gut microbiota impacts these circadian rhythms, yet the underlying mechanisms are largely unknown. We have demonstrated that the gut microbiota is essential for the rhythms of histone acetylation, which suggested that epigenetic regulation is a key mechanism by which the microbiota impacts host circadian pathways. In ongoing work, we are identifying epigenetic modifications that have microbiota-dependent rhythms and studying their functions.
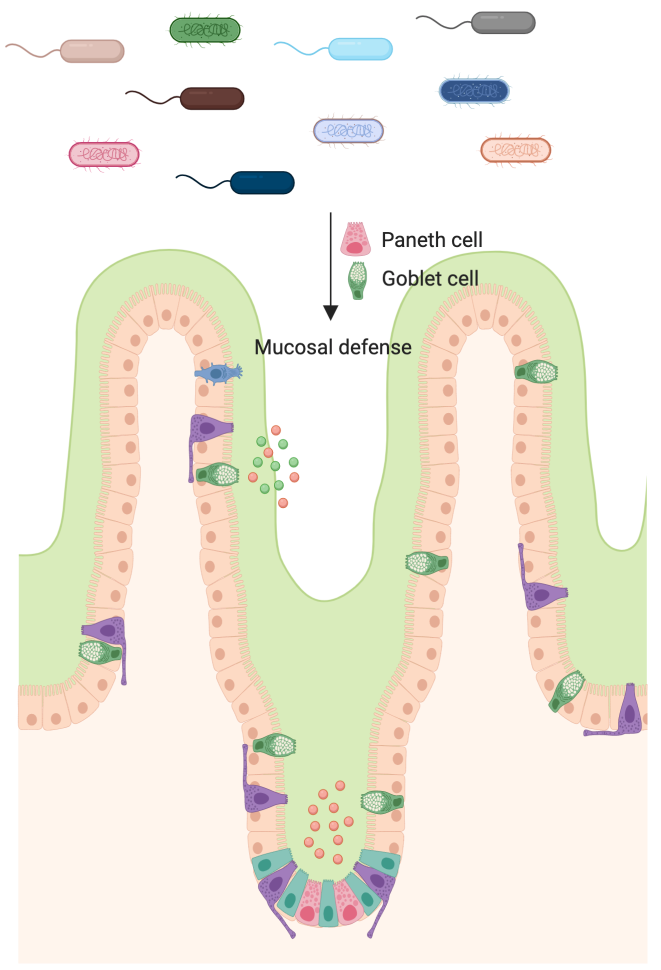
Microbiota-dependent epigenetic regulation of mucosal barrier functions
A harmonious mutualistic environment is critical for both the intestinal epithelium and bacteria to perform their metabolic reactions. However, excessive or abnormal host-microbial interactions pose a threat to metabolic health instead. Intestinal epithelial cells limit bacterial attachment and invasion by (1) forming a physical barrier with tight junctions; (2) secreting mucins which form the mucus layer that physically separates bacteria from host; and (3) secreting anti-microbial proteins that kill bacteria or restrict their growth. Mucins and antimicrobial proteins are synthesized and secreted mainly by two lineages of specialized epithelial cells: Paneth cells and Goblet cells. Despite the importance of these secretory cells in mucosal defense, how their activities are regulated remains largely unknown. In ongoing work, we are exploring epigenetic mechanisms by which the microbiota regulates these secretory cells to impact mucosal barrier functions.